Branchiomyces: What Kind of Fungus Attacks Fish in Warm Water?
Branchiomyces in Koi
Written by Dr Erik Johnson
Thursday, 03 April 2008 18:41
ABSTRACT: BRANCHIOMYCES
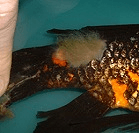
Branchiomyces is an aggressive gill fungus. It will kill fish by adulterating their gill tissue. The fungus compromises vascular supply to gill tissue and the affected areas die and slough out leaving gaping sockets in the gill arches. The fungus relies on high organic loads, high Nitrate levels, crowding, and HEAT to activate, and kill fish.
- Hot water organism
- Requires high organics
- Controlled wth Formalin
Recognizing Branchiomyces
Fish will linger at surface and die in three days, sometimes one by one, starting with the smallest fish.
When you examine the fish, you will notice the gills are eroded and there are necrotic sections in the gill tissues, grossly.
Special stains will show starlike fungal elements, but this is not critical because treatment is simple.
Limiting Losses from Branchiomyces:
Cool water will slow the fungal infection down and save fish lives, it REQUIRES hot water to thrive and is most common in intensive conditions where fish are crowded, there are high organic loads and the water is WARM.
- Cool water carries more oxygen and eases the metabolic furnace of the fish.
- Increasing aeration WITHOUT increasing exertion to the fish (e.g. spraybars) may be life saving.
Controlling Branchiomyces:
Formalin is very effective. For more information on how to use the Formalin or refer to the book.
Expected results:
Amazingly, when encountered, Branchiomyces is rapidly cleared.
The pond should be cleaned, organics in the filter and in the biofilm should be scrubbed or removed and the water should be cooled, which can and will be accomplished in part by the frequent water changes mandated by the Formalin treatment.
Fish will be noticeably perkier and feel better the day after the water is cooled and the Formalin is applied.
Because the gills are already compromised, no Formalin treatment should last longer than 2 hours.
Thanks!
—
Best Regards,
“Doc Johnson”
PS: This infection occurred in my own collection. This is first hand experience. I am glad I had my microscope and some “ins” with folks at University of Georgia to be able to figure out “What kind of super fungus prefers HOT water?” lol
WHAT FOLLOWS ARE MY NOTES PUTTING THIS SYNOPSIS TOGETHER
3. Anonymous. 1973. Fish diseases and their control. FAO Aquacult. Bull. 6(1):1618. An occurrence of a Branchiomyces sanguinis infection is described in fingerling striped bass reared at a fish hatchery in Arkansas. Repeated treatment with 25 ppm formalin relieved the infection. Tilling and drying of the bottom of the affected pond prevented recurrence of the disease during the following 3 years.
49. Meyer, F. P., and J. A. Robinson. 1973. Branchiomycosis: a new fungal disease of North American fishes.
Prog. FishCult. 35:7477. Mortality of fingerling striped bass at a fish hatchery in Arkansas was attributed to a gill disease identified as “gill rot” caused by the fungus Branchiomyces sanguinis. The affected gill tissues became necrotic and were white or brownish due to impaired circulation. Often necrotic tissues sloughed off, leaving denuded areas on thegill arches. Infected fish became extremely weak, showed respiratory distress, lost their equilibrium, and died. An initial pond treatment of formalin at 15 ppm, followed by applications of 25 ppm, stopped mortality. Repeated treatments were required to control the infection. The authors recommended that infected fish be destroyed and ponds sterilized before draining because branchiomycosis sometimes reappears if ponds are not thoroughly dried.
Diseases of Aquatic Organisms
DAO 31:233-238 (1997)
Abstract
Branchiomyces-like infection in a cultured tilapia (Oreochromis hybrid, Cichlidae) I. Paperna1,*, M. Smirnova21Department of Animal Sciences, Faculty of Agriculture of the Hebrew University of Jerusalem, Rehovot 76100, Israel 2The Central Fish Health Laboratory, Nir David 19150, Israel *E-mail: paperna@agri.huji.ac.il
ABSTRACT: A Branchiomyces-like fungus was identified from histologically examined gills of juvenile red tilapia Oreochromis niloticus × O. mossambicus and green tilapia O. niloticus × O. aureus obtained from intensive fish farms in Israel. Hyphae embedded in the gill tissue contained undivided and sporulating stages. In one instance infection appeared to be subclinical, limited to one or a few loci in the gill filament and causing only localized tissue damage. In the second case, infection, which was severe and caused extensive tissue damage, was identified in fish from a stock suffering massive mortality which resulted in the loss of 85% of the pond fish.
KEY WORDS: Branchiomyces · Gills · Oreochromis hybrid ·
Cichlidae · Mortality · Aquaculture · Israel
Published in DAO Vol. 31, No. 3 (1997) on December 30 ISSN: 0171-8630. Copyright Inter-Research, Oldendorf/Luhe, 1997
http://aquaweb.pair.com/DBWS/mycotic.html#branchyo
Branchiomycosis (Gill rot)
A) Caused by two species Branchiomyces sanguinis and B. demigrans.
B) Primarily a problem in carp and eels.
C) Affected fish usually show respiratory distress. There is prominent gill necrosis caused by thrombosis of blood vessels in the gills. Histologically the identification of nonseptated branching hyphae with an intrahyphal eosinophilic round body (apleospores) in and around blood vessels of the gill is diagnostic.
D) The disease occurs most commonly in ponds with abundant organic matter, and high ammonia levels.
Usually warm water temperatures (20-25øC) bring about the disease.
Use of DiQuat:
Another chemical is the herbicide Diquat. It has been used at a concentration of 8.4 to 16.8 ppm of the formulated material, or 2 to 4 ppm on the basis of active ingredient (Diquat cation).
Several chemicals have been used to treat BGD in hatchery salmonids. The most widely used are quaternary ammonium compounds, such as benzalkonium chlorides, available as Hyamine 1622 (98.8% active ingredient) and Hyamine 3500 (50% active ingredient). Another compound of this type is Roccal, available in 10% or 50% concentrations. Benzalkonium chlorides are used in concentrations of 1 to 2 ppm (calculated on the basis of the active ingredient) as a 1h bath or continuous flow treatment. Caution is necessary because the margin of safety is narrow, particularly in soft water.
None of these chemicals are approved by the U. S . Food and Drug Administration for disease control in food fishes. Efforts are under way to have chloramineT registered as a treatment for BGD. This compound was found effective for BGD when used at 8.5 ppm for I h (From 1980). In hatchery tests conducted by the National Fish Health Research Laboratory, one treatment effectively controlled BGD when it was given early in an outbreak. However, two or more treatments are required for advanced outbreaks.
When BGD, caused by C. columnaris, occurs in pondfish, external treatments for the control of columnaris disease are used.
The treatment with 8.5 ppm Diquat described above has been reported to be effective. Diquat, copper sulfate at 0.5 ppm, and potassium permanganate at 2 to 4 ppm can be added to ponds and allowed to dissipate over time. If copper sulfate or potassium permanganate are used, treatment levels may have to be adjusted, depending on water chemistry. In soft water, 0.5 ppm copper sulfate may be toxic; and if pondwater is high in organic material, potassium permanganate concentrations must be increased (Rogers 1971; Jee and Plumb 1981).
Fungal Diseases of Fish
Ruth Ellen Klinger and Ruth Francis-Floyd
Fungi are a group of organisms called heterotrophs that require living or dead matter for growth and reproduction. Unlike plants, they are incapable of manufacturing their own nutrients by photosynthesis. Fungi are present everywhere–in saltwater or fresh water, in cool or warm temperatures. In most cases, fungi serve a valuable ecological function by processing dead organic debris. However, fungi can become a problem if fish are stressed by disease, by poor environmental conditions, receive poor nutrition, or are injured. If these factors weaken the fish or damage its tissue, fungus can infest the fish. Fungi can also prevent successful hatching when it invades fish eggs.
Fungi are grouped by the morphology of various life stages. All fungi produce spores–and it is these spores which readily spread disease. The fungal spore is like a seed which is resistant to heat, drying, disinfectants and the natural defense systems of fish. The three most common fungal diseases are discussed here. They are known as Saprolegniasis, Branchiomycosis, and Ichthyophonus disease.
BRANCHIOMYCOSIS
Branchiomyces demigrans or “Gill Rot” is caused by the fungi Branchiomyces sanguinis and Branchiomyces demigrans .
Branchiomycosis is a pervasive problem in Europe, but has been only occasionally reported by U.S. fish farms. Both species of fungi are found in fish suffering from an environmental stress, such as low pH (5.8 to 6.5), low dissolved oxygen, or a high algal bloom. Branchiomyces sp. grow at temperatures between 57° and 95°F but grow best between 77° and 90°F. The main sources of infection are the fungal spores carried in the water and detritus on pond bottoms.
Disease Signs
Branchiomyces sanguinis and B. demigrans infect the gill tissue of fish. Fish may appear lethargic and may be seen gulping air at the water surface (or piping). Gills appear striated or marbled with the pale areas representing infected and dying tissue. Gills should be examined under a microscope by a trained diagnostician for verification of the disease. Damaged gill tissue with fungal hyphae and spores will be present. As the tissue dies and falls off, the spores are released into the water and transmitted to other fish. High mortalities are often associated with this infection.
Management and Control
Avoidance is the best control for Branchiomycosis. Good management practices will create environmental conditions unacceptable for fungi growth. If the disease is present, do not transport the infected fish. Great care must be taken to prevent movement of the disease to noninfected areas. Formalin and copper sulfate have been used to help stop mortalities; however, all tanks, raceways, and aquaria must be disinfected and dried. Ponds should be dried and treated with quicklime (calcium oxide).
